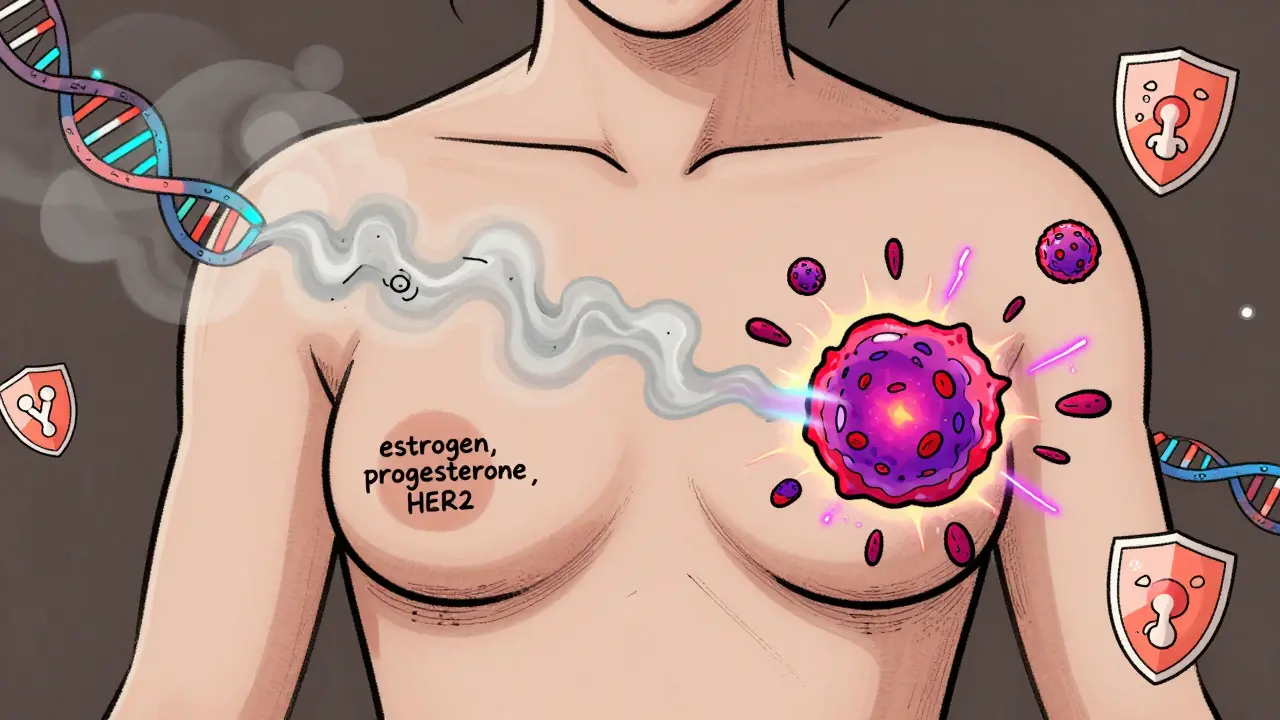
Triple-negative breast cancer treatment has evolved beyond chemotherapy. Learn how immunotherapy, PARP inhibitors, antibody-drug conjugates, and personalized vaccines are changing survival rates and reducing toxicity in 2026.
read more
Type 1 diabetes requires lifelong insulin therapy. Learn the key symptoms, how it's diagnosed with blood tests and autoantibodies, and the latest insulin delivery options including pumps and hybrid closed-loop systems.
read more
Vitamin E can increase the bleeding risk when taken with warfarin, especially at doses above 400 IU daily. Even if INR levels stay stable, dangerous bleeding can occur. Always consult your doctor before taking any supplement while on anticoagulant therapy.
read more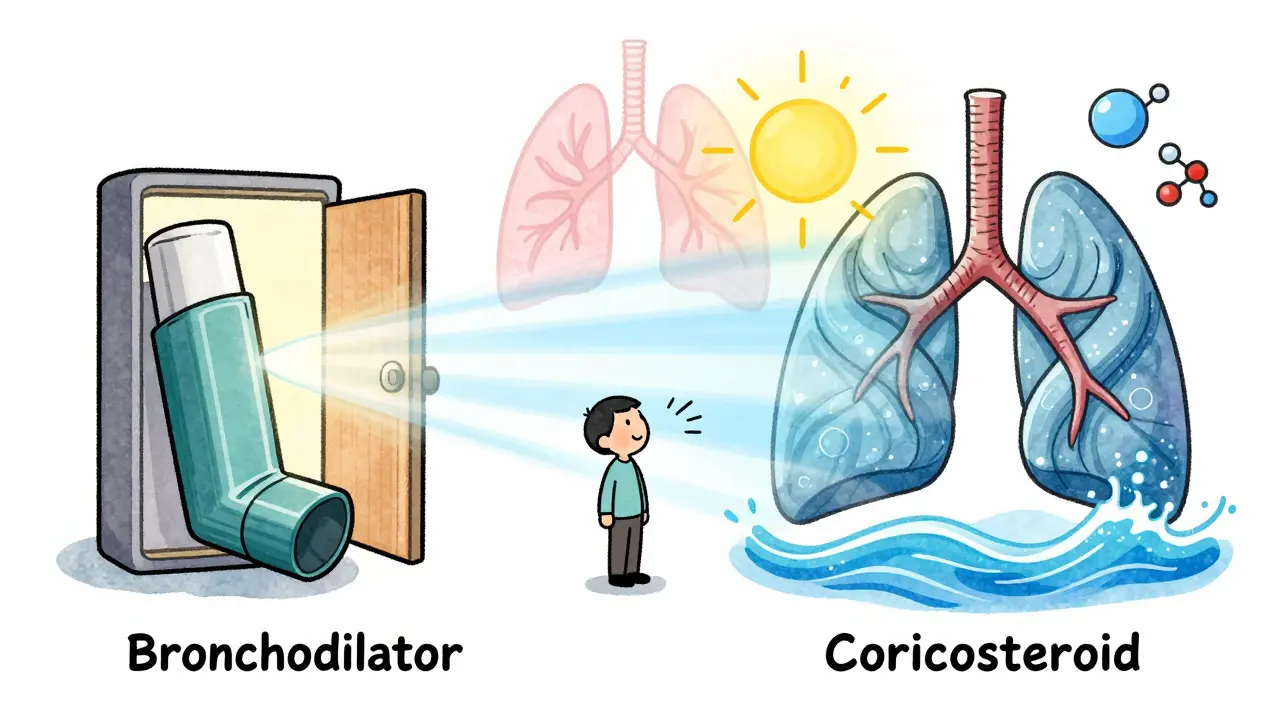
Bronchodilators open airways for quick relief; corticosteroids reduce inflammation for long-term control. Used together correctly, they manage asthma and COPD effectively. Learn how to use them in order and avoid common mistakes.
read more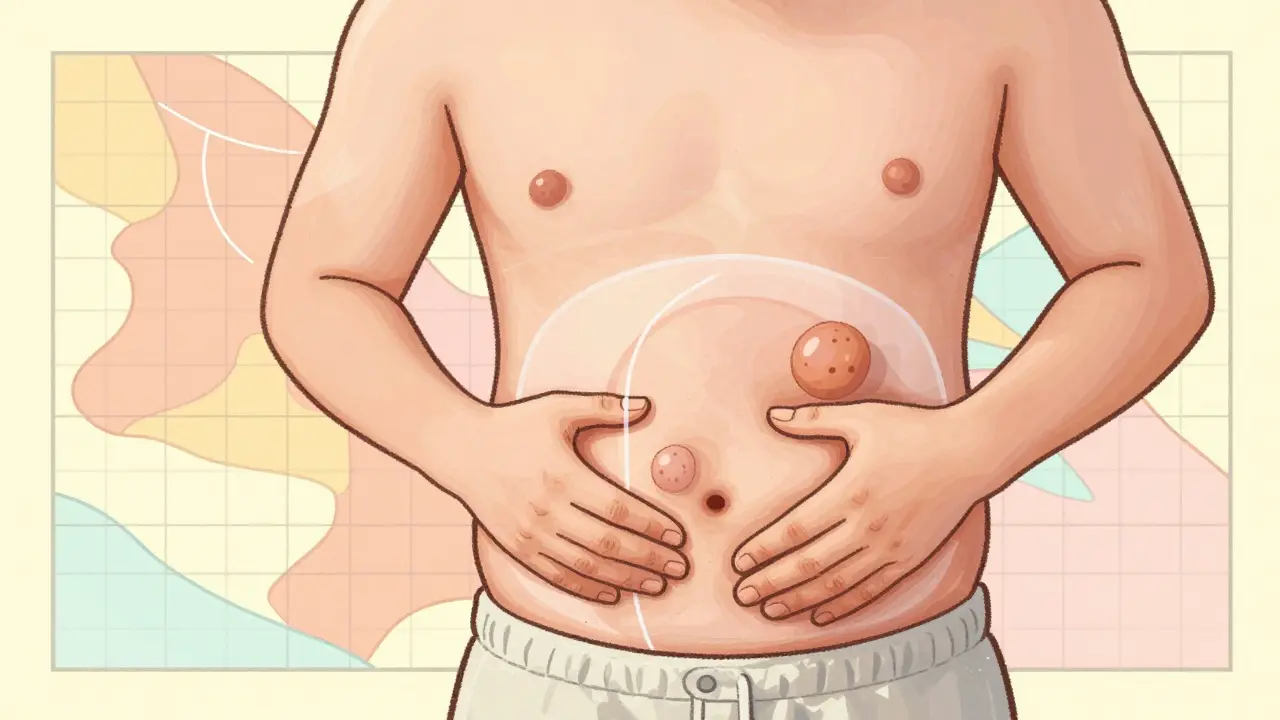
Lipodystrophy and bruising from insulin injections are common but often ignored. Learn how to spot lumps, prevent tissue damage, and stabilize your blood sugar with proper injection techniques.
read more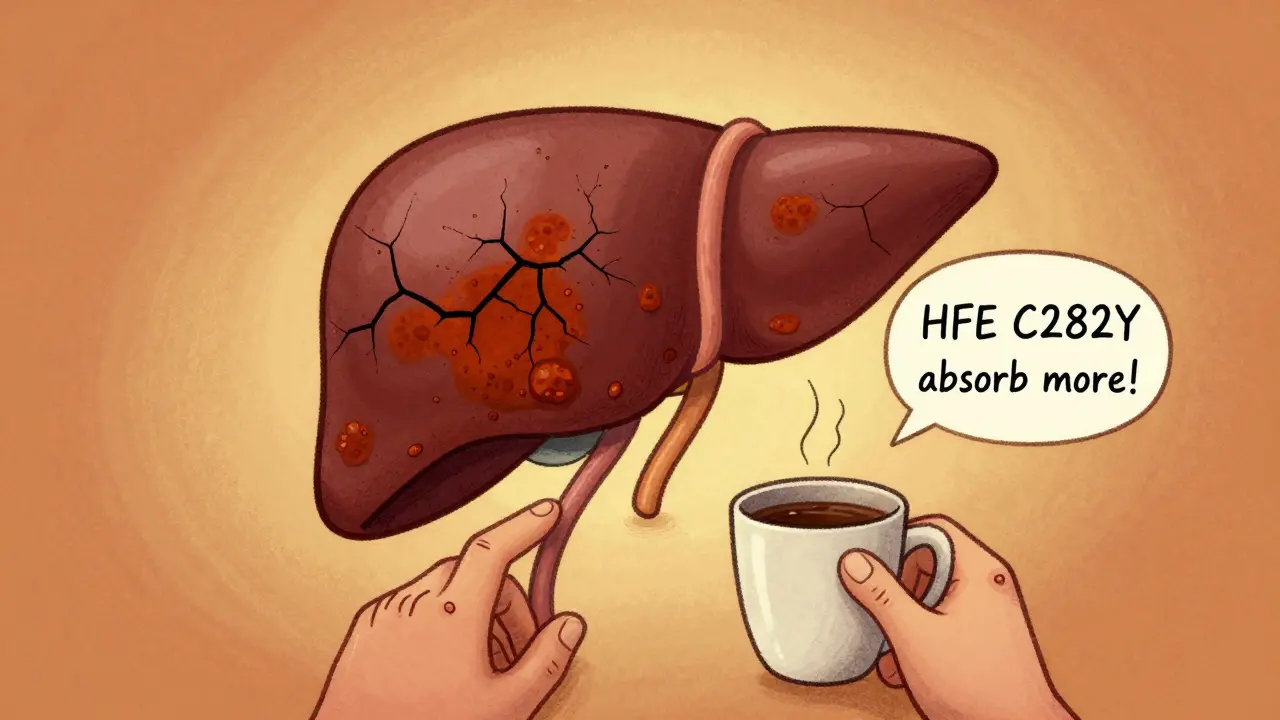
Hemochromatosis is a genetic iron overload disorder that damages the liver, heart, and pancreas. Phlebotomy-regular blood removal-is the only proven treatment that can prevent organ failure and restore normal life expectancy.
read more
Anti-Xa monitoring for LMWH isn't routine-it's for emergencies. Learn when unexpected bleeding, clots, or kidney issues mean you need this test to stay safe.
read more
Crossover trial designs are the standard method for proving bioequivalence between generic and brand-name drugs. They reduce sample sizes, improve accuracy, and are required by the FDA and EMA for most applications. Learn how they work, when to use replicate designs, and why washout periods matter.
read more
In vivo and in vitro bioequivalence testing are key to approving generic drugs. In vivo uses human subjects to measure drug absorption; in vitro uses lab tests like dissolution. Each has specific uses based on drug type, safety, and regulatory rules.
read more
The DSCSA track-and-trace system uses unique serial numbers and electronic verification to stop counterfeit drugs from reaching patients. Learn how it works, why it matters, and what’s changed since 2024.
read more
Learn the safest, most reliable ways to keep insulin, Mounjaro, and other refrigerated medications cold while traveling in 2025. From TSA rules to top coolers and real traveler tips.
read more
Learn how to talk to patients about generic medications in a way that builds trust, improves adherence, and saves money-without sounding dismissive or robotic.
read more