
Explore the risks of switching child medications to generics. Learn about bioequivalence gaps, narrow therapeutic index drugs, and how insurance formulary changes impact pediatric health.
read more
Discover how the FDA monitors generic drug safety after approval using FAERS, the Sentinel Initiative, and rigorous factory inspections to ensure public health.
read more
Save hundreds on generic Depakote (divalproex sodium). Compare prices from GoodRx, SingleCare, and online pharmacies. Learn safe ways to buy cheap medication online in 2026.
read more
Learn how pharmacy substitution laws vary by state, impacting generic and biosimilar drug access. Understand mandatory vs. permissive rules, patient rights, and key steps to navigate medication changes safely.
read more
Find the safest and cheapest ways to buy generic Cymbalta (duloxetine) online in 2026. Compare prices from GoodRx, RedBox Rx, Amazon, and Canadian pharmacies with safety tips.
read more
Learn how to safely buy Tofranil online in 2026. We cover telemedicine prescriptions, trusted pharmacies, generic vs. brand costs, and safety checks to avoid scams.
read more
Learn how to effectively report medication side effects to your doctor using data, diaries, and the 5 Ws framework to ensure your health concerns are taken seriously.
read more
Learn how to buy Aleve online with a full guide to the best retailers, pricing tips from Walmart, and delivery options from Target and Walgreens.
read more
Compare the costs of brand biologics versus biosimilars. Learn why price gaps exist, how much you can save, and the factors influencing drug pricing in 2026.
read more
Achieving SVR cures Hepatitis C, but does it eliminate liver cancer risk? Learn who still needs HCC surveillance, the role of liver fibrosis, and current global guidelines.
read more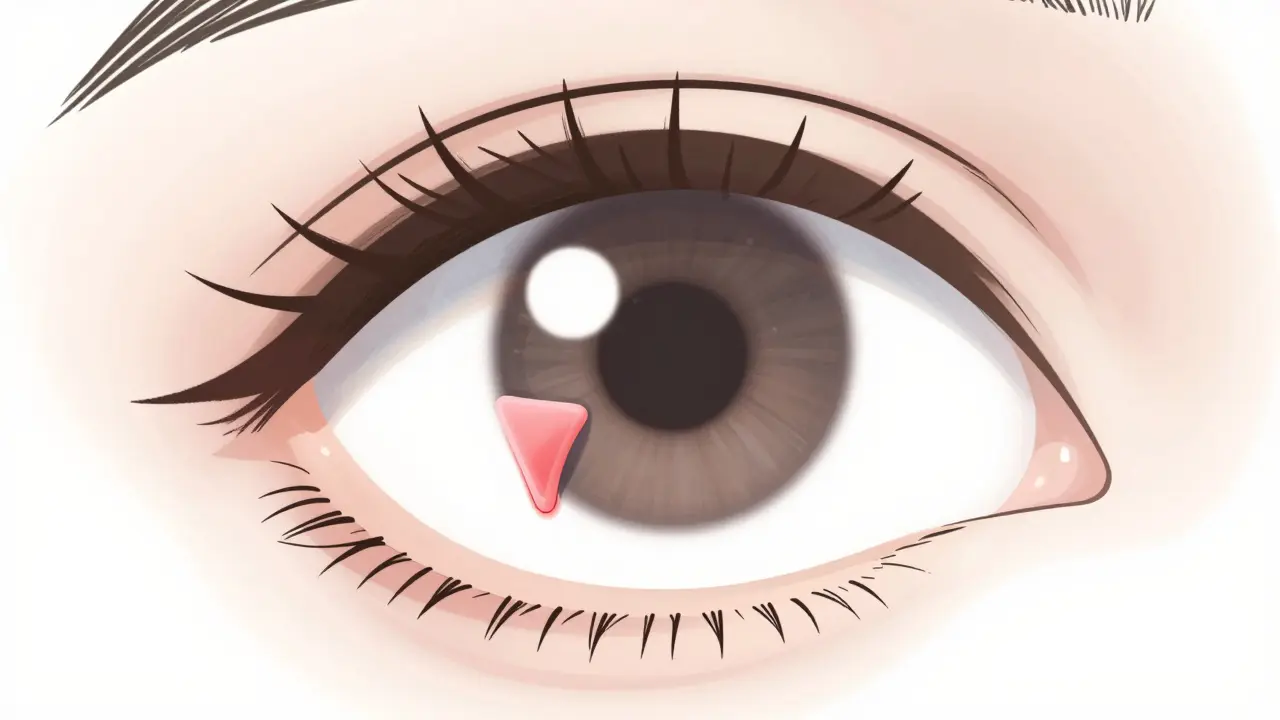
Learn about Pterygium (Surfer's Eye), how UV sun exposure drives its growth, and the most effective surgical options to prevent recurrence and save your vision.
read more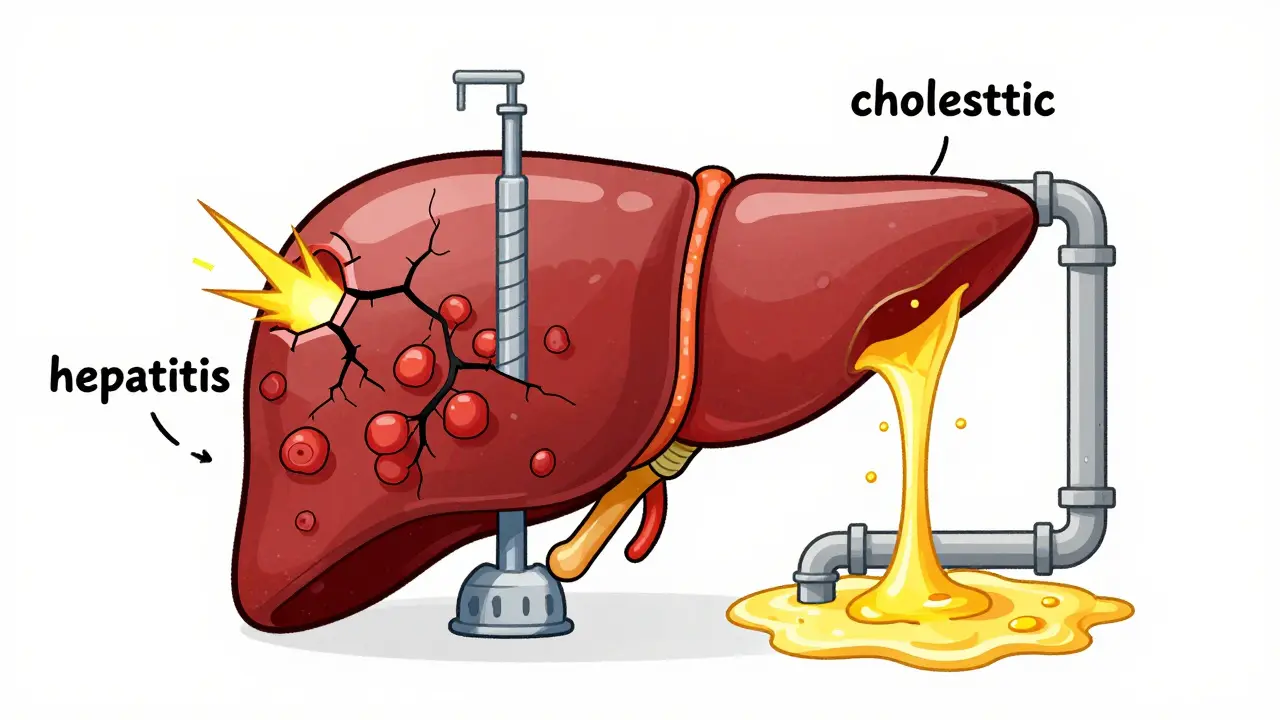
Learn about antibiotic-related liver injury, the difference between hepatitis and cholestasis patterns, high-risk drugs like amoxicillin-clavulanate, and prevention tips.
read more